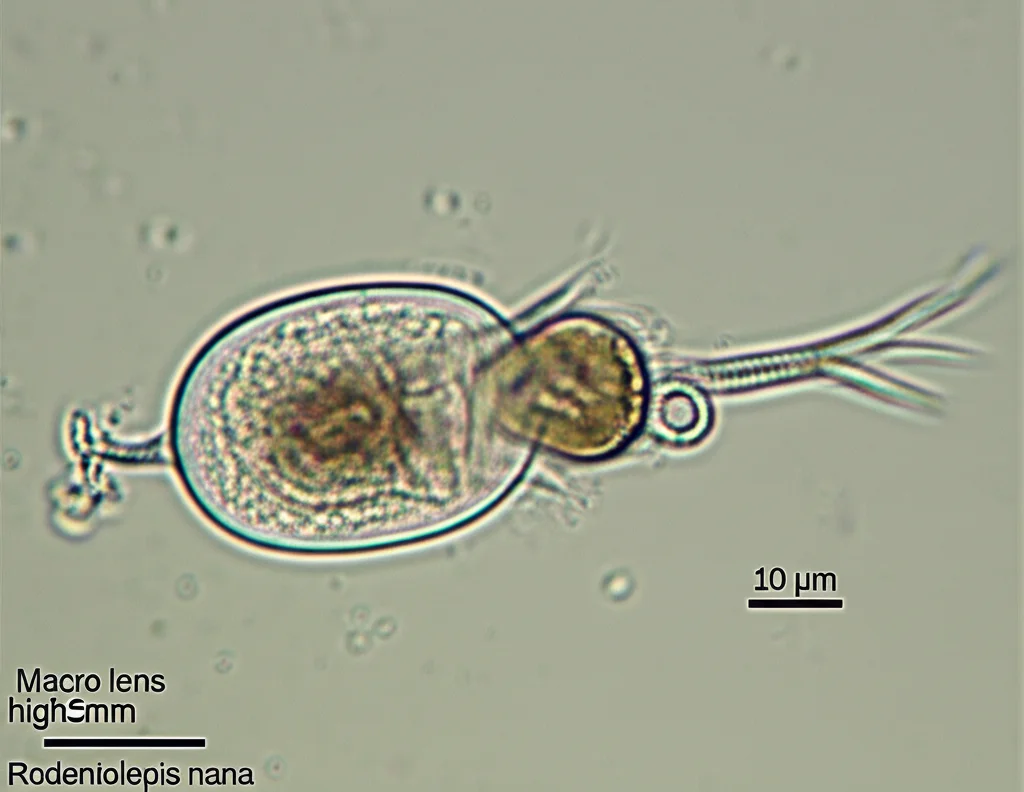
Unpacking the Dwarf Tapeworm: What We Learned from Refugee Health Screening
Hey There, Let’s Talk About Health Screening!
So, you know how health is super important, right? Especially when people are arriving in a new place, perhaps having gone through some tough times. That’s where services like Respond in London come in. They’re set up specifically to help people seeking asylum and refugees get settled and healthy. Part of what they do is offer health screening to catch common issues early on. And let me tell you, sometimes what you find is pretty interesting, even if it’s tiny!
We’ve been taking a look back at the data from the Respond service, specifically focusing on something called Rodentolepis nana. Don’t let the fancy name scare you; it’s actually the most common type of tapeworm that infects humans. It’s often called the “dwarf tapeworm” because, well, it’s small! We wanted to dig into how often we were seeing this little guy in the folks coming through for screening and how well the usual treatment works.
Meet the Dwarf Tapeworm: Tiny but Troublesome
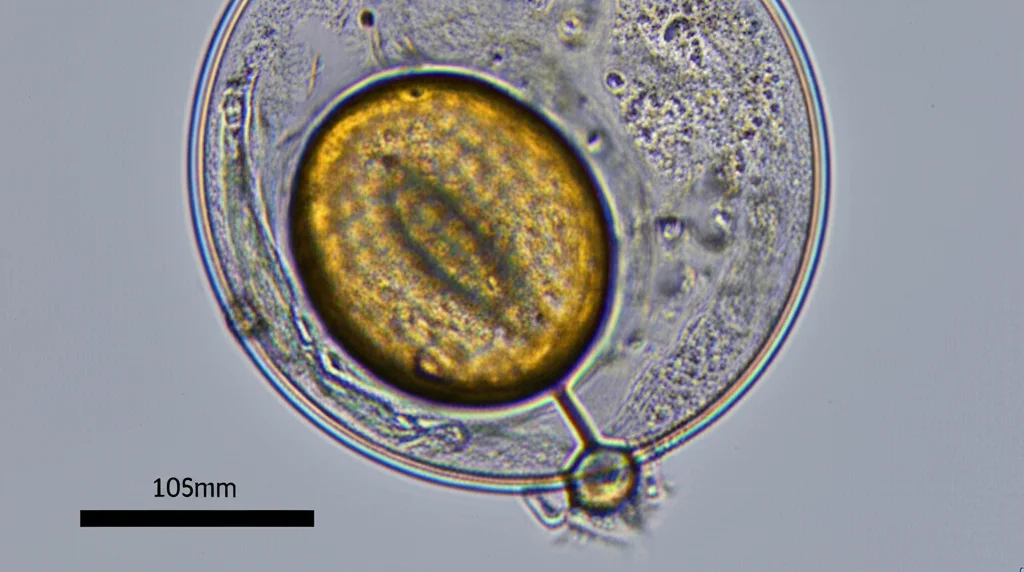
Imagine a tapeworm that’s only about 2–4 cm long and just a millimeter wide. That’s R. nana for you! What makes it unique among human tapeworms is that it can complete its whole life cycle inside just one person. No need for a middleman host, though it can also hang out in rodents and some insects.
While it can be passed from animals, the most common way it spreads is directly from one person to another. These little tapeworms produce eggs that are infectious right away after they leave the body. This makes it super easy for it to spread within families or in crowded living situations. The eggs are pretty tough too, surviving up to 10 days outside the body, ready to contaminate food or water.
And here’s a slightly unsettling bit: R. nana is the only human tapeworm that can cause “autoinfection.” This means the eggs can hatch inside your gut before they even leave your body, or you can reinfect yourself if eggs get on your hands. So, even though each individual tapeworm only lives for about 4–6 weeks, you can keep the infection going for years without even realizing it!
Why We Looked at This Group
R. nana is found all over the world, but it’s more common in places with temperate climates, lower socioeconomic status, crowded living conditions, and limited access to clean water and sanitation. Sound familiar? These are often the very challenges faced by people seeking asylum and refugees, both in their countries of origin and during their journeys. Studies have shown that parasitic infections are often more common in people migrating from high-prevalence areas compared to the general population in places like the UK.
While many people carrying R. nana don’t show any symptoms, those with a lot of worms can experience:
- Abdominal pain
- Diarrhoea
- Weakness
- Growth stunting (especially in kids)
In very rare cases, usually in people with weakened immune systems, it can even lead to more serious complications. Plus, it often comes along with other infections, particularly Giardia duodenalis, and might contribute to poor growth and nutrition when other infections are also present.
Given all this, screening for parasites like R. nana is often recommended for this population, though exactly how and when varies. The Respond service makes it part of their integrated health assessment, offering testing and treatment right there.
How We Did It
We looked back at all the positive R. nana results from stool samples collected at the Respond service between April 2016 and July 2023. How did we find it? Well, the lab uses a couple of methods:
- Microscopy: Looking at concentrated stool samples under a microscope to spot the tiny eggs.
- Molecular Testing: More recently, they introduced a test called Novodiag® Stool Parasite assay (NSP), which detects the parasite’s DNA. It’s a bit like a super-sensitive search party looking for genetic fingerprints. Even when they started using the NSP, they still confirmed positive results with microscopy.
We then matched these positive results back to the patient’s records to see who they were, where they were staying, and if their family members were also tested.

What We Found: The Numbers Tell a Story
Out of 1797 patients who provided a stool sample for screening at Respond during our study period, 54 (that’s about 3%) tested positive for R. nana. This prevalence is actually pretty similar to what you might see in countries where the infection is common.
Here’s a quick snapshot of the patients who tested positive:
- Age: The median age was 15 years, with a big chunk (81%) being under 18. This fits with what we know about R. nana being more common in children.
- Sex: 70% were male, which generally reflects the demographics of the Respond cohort.
- Time in UK: On average, they had been in the UK for about 5 months when the sample was taken.
- Accommodation: They were staying in various places – hotels/hostels, foster care, or other types of accommodation.
- Origin: Patients from Afghanistan and Eritrea were seen more often in our positive group compared to their representation in the overall Respond population.
We also saw that having other tummy bugs was really common among those with R. nana. Over half (52%) had at least one other parasite or infection, like Giardia or Helicobacter pylori. This makes sense, as they often spread the same way – through contaminated food or water.
Family Ties and Treatment Troubles
Now, here’s a really important finding: R. nana loves to stick together in families. For the patients whose family members were also tested, a significant number (41%) had at least one family member who was also infected! This happened in 4 out of the 5 families where multiple members were tested. This strongly suggests that transmission is happening within households.
When someone tested positive, they were given treatment – usually a single dose of praziquantel, which is the standard medication. Patients were asked to come back for a follow-up stool sample about a month later to check if the infection was gone. This is where things got a bit tricky.
Out of the patients who *did* provide a follow-up sample (not everyone did, which is a challenge in itself for this mobile population), a surprisingly high number – 43% – still tested positive for R. nana! This means the treatment didn’t work, or they got reinfected quickly. Interestingly, all the patients who failed treatment were children. Many were in large family groups, and some had other family members who were also infected.

What Does It All Mean?
Seeing a 3% prevalence of R. nana in this group confirms that it’s a real issue for people seeking asylum and refugees coming to the UK, likely linked to the challenging conditions they may have faced. The fact that it often comes with other infections highlights the broader risks from poor sanitation and hygiene.
The strong clustering within families isn’t surprising, given shared living spaces and the easy person-to-person spread of R. nana eggs. What was particularly interesting was that in the families we looked at, it seemed to be siblings infecting each other more than parents infecting children. This might point towards direct behavioral transmission (like hands not being washed properly) being a big factor, perhaps even more so than just sharing the same food or water.
The high rate of treatment failure (43% in those with follow-up) is a bit concerning. It could be that the medication didn’t fully clear the infection, or maybe they got reinfected very quickly. Given the crowded living conditions and family clustering, reinfection seems like a very likely culprit. Imagine clearing the infection, only to pick it up again from a family member or the shared environment within weeks! Poor treatment adherence (not taking the medication properly) could also play a role, as we saw one patient clear the infection only after the treatment was supervised.
While praziquantel is the go-to treatment, maybe alternative medications like nitazoxanide or albendazole could be considered, although more research is needed to compare them directly to praziquantel.
Moving Forward: What Can We Do?
Based on what we found, it seems pretty clear that testing for R. nana in people seeking asylum and refugees is a good idea. Catching these infections early can prevent ongoing transmission and potential health issues, especially growth problems in children.
Because it spreads so easily within families, we think it makes a lot of sense to test whole families together. And if one person is positive, maybe even consider treating the whole family unit at the same time, even if some members tested negative initially. This could help break the cycle of reinfection.
Beyond treatment, tackling the root causes is key. This means better education on hygiene, improved sanitation, and less crowded living conditions. These are big challenges, especially for this population who often face barriers to healthcare like language issues, frequent moves, and stigma. Providing support to ensure people take their medication properly is also important, perhaps through supervised doses or extra counselling.
Of course, our study had some limitations. It was a look back at existing data, and we couldn’t definitively say *how* transmission was happening (genomic testing could help with that) or why treatment failed (was it reinfection, adherence, or something else?). Not everyone came back for follow-up testing, which also makes it harder to get the full picture of treatment success.
But overall, our findings highlight that R. nana infection is present in this population, it clusters in families, and treatment isn’t always a one-shot deal. Taking a proactive approach to screening and considering family-based strategies seems like the way to go to help these individuals stay healthy.
Source: Springer