Your Body’s Inner Fire: How Temperature, CMA, and PARK7 Control Fat Metabolism
The Warm Truth About Your Brown Fat
You know how temperature affects us? Especially our metabolism? Well, there’s this special kind of fat called brown adipose tissue, or BAT, and it’s a big deal for burning energy and keeping us warm, especially when it’s chilly. It does this with a neat protein called uncoupling protein 1 (UCP1), which helps generate heat instead of storing energy. This process significantly boosts our overall energy expenditure.
But let’s be real, most of us aren’t hanging out in the cold all the time. We live in comfy, thermoneutral spots – temperatures where our body doesn’t need to work hard to stay warm. And guess what? When we’re in these cozy conditions, our BAT gets a bit lazy. It goes through this process called “whitening,” where it starts looking and acting more like regular white fat, storing energy instead of burning it. This involves accumulating big fat droplets and the energy-burning machinery, like mitochondria, starts to decline.
A key player in BAT’s energy-burning power is a protein called PGC1α. It’s like a master switch for metabolism, especially for making BAT work. When it’s cold, PGC1α levels go up, and BAT gets busy. But under thermal stress, like being in a warm environment, PGC1α levels drop. The big question has been: *how* exactly does the body get rid of this crucial protein when it’s not needed for heat? That was the mystery we set out to solve.
Unmasking the Culprit: CMA Steps In
So, we started poking around in cells and brown fat tissue to figure out how PGC1α disappears under thermal stress. We knew proteins are usually broken down by something called the proteasome, sort of like the cell’s trash compactor. We tried blocking the proteasome at warmer temperatures, but guess what? It didn’t fully stop PGC1α from degrading. Hmm. This told us another system was involved.
What else cleans up proteins in the cell? Autophagy! This is a process where the cell packages up stuff it wants to get rid of and sends it to the lysosome, the cell’s recycling center. When we blocked autophagy using a common inhibitor, *boom*! PGC1α levels stayed high, even at warmer temps. Okay, so autophagy is definitely involved in getting rid of PGC1α under thermal stress.
But autophagy isn’t just one thing; there are different types. We looked at the main ones: macro-autophagy and micro-autophagy. Turns out, messing with the key players in those didn’t really affect PGC1α levels. Then we looked at chaperone-mediated autophagy, or CMA. CMA is pretty unique because it selectively degrades specific proteins that have a particular sequence (called a KFERQ-like motif). It uses a chaperone protein called HSC70 to recognize these proteins and deliver them to the lysosome via a special receptor protein on the lysosomal surface called LAMP2A.
When we messed with LAMP2A levels – knocking it down or overexpressing it – we saw a direct impact on PGC1α. Knocking down LAMP2A increased PGC1α, and overexpressing it decreased PGC1α. This was a big clue! It strongly suggested that CMA, using LAMP2A, is the specific type of autophagy responsible for degrading PGC1α under thermal stress. We even found the specific KFERQ-like motifs on PGC1α that CMA likely targets.

CMA’s Impact on BAT Function: A Mouse Story
Alright, lab dishes are one thing, but what about a whole living system? We wanted to see what happens when we mess with CMA specifically in BAT in mice. We used a neat genetic trick to knock down Lamp2a (the gene for LAMP2A) just in their brown fat. We then housed these mice in a thermoneutral environment to mimic human living conditions.
And guess what? At thermoneutrality, these mice with reduced CMA activity in their BAT were doing great metabolically compared to control mice! They gained less weight, had less fat mass, and were burning more energy, especially during their active phase. They even showed improved sensitivity to insulin, which is super important for preventing diabetes. Their BAT looked healthier too – the fat cells were smaller, and there was more of that energy-burning UCP1 protein. We also saw higher levels of both PGC1α and UCP1 protein in their BAT.
These data were pretty compelling. They suggested that under thermoneutral conditions, CMA is actively degrading PGC1α and UCP1, contributing to BAT whitening and metabolic slowdown. Inhibiting CMA in BAT seemed to keep it more active and metabolically beneficial, even in the warmth.
Enter PARK7, the Temperature Sensor
This got us thinking: if CMA is so important for BAT function under thermal stress, how is its activity being regulated by temperature in the first place? We went on a scientific treasure hunt, analyzing all the proteins in BAT from mice housed at different temperatures (cold, room temp, and thermoneutrality). We combined this with tools that predict how proteins might interact with each other, specifically looking for proteins that might talk to LAMP2A.
And one protein kept popping up as a potential key player: PARK7. You might have heard of PARK7 because mutations in its gene are linked to early-onset Parkinson’s disease, but it’s also known to be involved in various metabolic processes. Interestingly, we saw that the level of PARK7 protein in BAT decreased significantly when mice were housed at thermoneutrality compared to room temperature.
Further experiments confirmed that PARK7 does indeed interact with LAMP2A. It also interacts with HSC70, the chaperone that brings proteins to LAMP2A for CMA degradation. It looks like PARK7 is like a gatekeeper or a competitor. It seems to compete with HSC70 for binding to the same spot on LAMP2A. When PARK7 is around and bound to LAMP2A, it might block HSC70 from bringing substrates, thus slowing down CMA. When PARK7 levels drop (like at warmer temps), there’s less competition, allowing HSC70 to bind LAMP2A more easily and ramp up CMA activity. This interaction between PARK7 and HSC70 even seemed stronger at warmer temperatures, suggesting a complex temperature-sensing mechanism.
PARK7’s Role in BAT Whitening: The Flip Side
To really nail down PARK7‘s role in this whole process, we created special mice where the Park7 gene was specifically knocked out *only* in their BAT. We called these mice Park7ΔUcp1. When we housed these mice under standard conditions, they seemed fine. But when we put them in a thermoneutral environment, things changed dramatically.
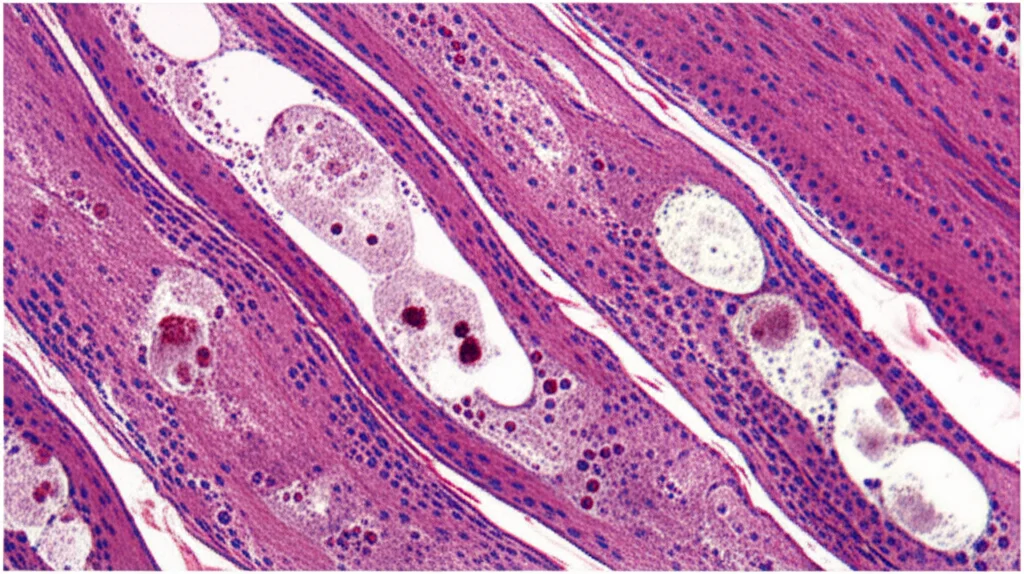
Compared to control mice, the Park7ΔUcp1 mice gained significantly more weight and fat mass. They burned less energy overall and had impaired glucose and insulin tolerance – signs of metabolic dysfunction. Their BAT was clearly undergoing accelerated “whitening.” Microscopic analysis showed larger fat droplets, fewer and degenerated mitochondria (the powerhouses of the cell), and increased signs of inflammation.
Interestingly, while the *genes* for thermogenic proteins like PGC1α and UCP1 were actually *increased* in the Park7ΔUcp1 BAT (the cell was trying to make more), the *protein* levels of PGC1α and UCP1 were *decreased*. This is a crucial point: it wasn’t a problem with making the proteins, but with them disappearing *after* they were made. This fits perfectly with the idea that losing PARK7 somehow *enhances* the degradation system – CMA. So, it seems that in the absence of PARK7 at thermoneutrality, CMA goes into overdrive, breaking down PGC1α and leading to BAT dysfunction and metabolic problems.
Putting it All Together: The CMA-PARK7-PGC1α Axis
So, we had two seemingly opposite results: blocking CMA was good for BAT at TN, and losing PARK7 was bad (because it seemed to *increase* CMA). The ultimate test to confirm our model was to see what happens if we block CMA *in* the mice that are missing PARK7 in their BAT. Would it rescue the negative effects?
We injected the Park7ΔUcp1 mice with a virus that knocked down Lamp2a (inhibiting CMA) specifically in their BAT, and then housed them at thermoneutrality. And guess what? It was like hitting the reset button! These mice, which were missing PARK7 but had reduced CMA activity, gained similar weight and fat mass as the healthy control mice, and significantly less than the Park7ΔUcp1 mice with normal CMA. Their energy expenditure, glucose tolerance, and insulin sensitivity were largely restored to normal levels.
Looking at their BAT, the fat cells were smaller, inflammation was reduced, and mitochondrial function was improved. Crucially, the protein levels of PGC1α and UCP1 were increased again, just like in the mice where we only blocked CMA.
This pretty much sealed the deal. It confirmed that the negative metabolic effects seen in the Park7ΔUcp1 mice at thermoneutrality were indeed caused by the enhanced activity of CMA. The lack of PARK7 leads to increased CMA, which degrades PGC1α, causing BAT to whiten and energy metabolism to suffer.
Why This Matters
So, what’s the takeaway from all this? We’ve uncovered a pretty neat pathway: under thermal stress (like living in a warm house), the protein PARK7 decreases. This decrease allows the cellular cleanup crew, CMA, to become more active. CMA then specifically targets and degrades PGC1α, the master regulator of BAT’s energy-burning function. With less PGC1α, BAT loses its power, undergoes whitening, and contributes to metabolic slowdown and issues like weight gain and insulin resistance.
Why does this matter for us? Well, if we can understand *how* BAT loses its punch in warm conditions – conditions most humans live in – maybe we can find ways to keep it active. Targeting this CMA-PARK7-PGC1α pathway could be a potential strategy to preserve or regain BAT’s metabolic benefits in humans, which could be a neat way to help tackle obesity and related metabolic diseases. It sheds light on the complex ways our cells adapt to the environment and how these processes can impact our overall health.
Source: Springer